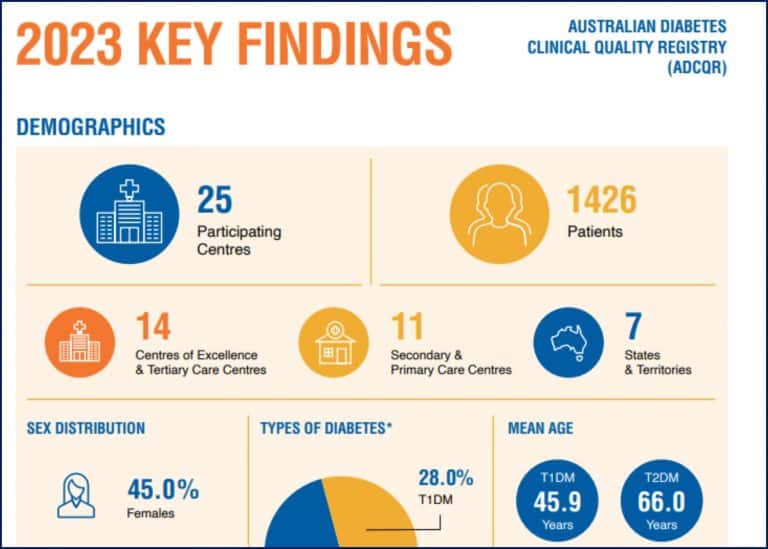
The Australian Diabetes Clinical Quality Registry (ADCQR)
(Formerly ANDA)
The Australian Diabetes Clinical Quality Registry (ADCQR) is the successor to the Australian National Diabetes Audit (ANDA) and its longitudinal counterpart (ANDA-L).
The ADCQR is a quality improvement initiative which serves to promote best practice and high-quality diabetes care. In line with the National Strategy for Clinical Quality Registries and Virtual Registries, the ADCQR aims to collect, monitor, and report on clinical quality outcomes to support improvements in clinical care and patient-centred outcomes. This will be achieved through benchmarked feedback on whether care across diabetes centres (and patient outcomes) align with that of their peers, as well as national standards.
In addition, the ADCQR aims to provide a longitudinal assessment of the effects of diabetes centres health care practices on patient treatment and outcomes through data linkage.
The Australian Commission on Safety and Quality in Health Care have identified diabetes as a priority area for development of a clinical quality registry due to the high burden of disease. Therefore, the data and datasets created as part of these activities within the institution, are of regulatory, clinical and community significance.
The ADCQR collects clinical indicators collected as part of standard care and include diagnoses, clinical and laboratory results, management methods/therapy and diabetes-related complications and comorbidities. Patient reported outcome measures are also collected capturing information on self- management and health service use. For more information about the ADCQR please visit https://www.monash.edu/medicine/sphpm/adcqr/home
To register an Expression of Interest and to be a part of the ADCQR, please email: adcqr@monash.edu.
Any questions from centres should be directed to Prof Sophia Zoungas at email: adcqr@monash.edu.
Download ADCQR Invitation letter here.
ADCQR ANNUAL REPORT
We can’t improve that which we don’t measure!
The Australian Diabetes Clinical Quality Register (ADCQR) welcomes participation from all diabetes centres around Australia.
ABOUT ADCQR
The ADCQR is a new national clinical quality registry with its foundations established from the Australian National Diabetes Audit (ANDA).
The ADCQR aims to monitor the quality (appropriateness and effectiveness) of health care for patients with diabetes by routinely collecting and analysing clinical performance data.
It aims to also provide clinicians, health service managers, patients and other stakeholders with ongoing, risk adjusted, benchmarked feedback on clinical practice and patient outcomes, to improve the standard of care for patients with diabetes.
ADCQR PARTICIPATION
To contribute to the ADCQR, your centre is required to register their interest by contacting the ADCQR Secretariat on [ADCQR email]. Your diabetes centre must nominate a local Principal Investigator (PI). The PI is responsible for site participation and compliance with ADCQR policies and procedures.
Getting Started
Once initial contact has been made, the ADCQR Secretariat will advise your centre on how to begin preparing for ADCQR data collection. ADCQR Central Management will help coordinate application submissions for local ethical and governance approval. ADCQR will provide all necessary resources to assist and train all relevant staff prior to data collection commencing.
There are three stages of setting up your diabetes centres as a site for ADCQR participation:
- Engagement of relevant site staff, departments and administration
- Human Research Ethics Committee (HREC) & local governance approvals
- Training site staff
Requirements for Participation
To participate in ADCQR your diabetes centre must be able to meet the following criteria:
- Ability to enter all eligible patients onto the database;
- Ability to nominate a PI to oversee and manage local data collection
- Ability to establish and maintain internal systems for data accuracy and timely data entry;
- Willingness to comply with the project protocol and HREC requirements;
- Willingness to sign a ADCQR Collaborative/Data Sharing Agreement
DATA COLLECTION
Your diabetes team will collect routine clinical information on patients attending your centre over a specified sampling period of every year (4-8 weeks during the months of May/June). This sampling period may be extended up until the censorship date of 31 August upon agreement between your site and the ADCQR Central Management Team to accommodate for any delays in ethics and governance approvals, and/or reliant on your site’s capacity to collect more data.
The information collected is part of your patients’ routine care. Information collected will include patient’s name, address, date of birth, sex, basic information about health status (height, weight, blood pressure), diabetes management,
complications, test results and medications. Your team may also choose to collect patient-reported outcomes about self- management of health and lifestyle, quality of life and well-being. Identifiable information will be collected for the purpose of patient follow up through data linkage to information held by Australian Commonwealth and State and Territory Government agencies, as well as other clinical registries and pathology databases.
The censorship date (latest date for data submissions into the ADCQR) to allow for analysis, and annual pooled reporting and site reporting is 31 August of every year.
DATA SECURITY & STORAGE
Your centre will be assigned a unique code by the ADCQR Secretariat. When collecting patient data for this activity, your site will be required to assign and document unique patient codes to secure patient identity. These patient codes linking patient personal details are required to be stored securely onsite and with the ADCQR Coordinating Centre. They will be stored
ANDA Background
The Australian National Diabetes Audit (ANDA) is a national clinical quality registry.
It is a centralised quality assurance activity aiming to improve the quality of care provided to people with diabetes.
ANDA is coordinated by Monash University in conjunction with the National Association of Diabetes Centres and participating primary, secondary and tertiary care diabetes centres across Australia.
ANDA collects data about people with diabetes treatments, complications, self-care practices and quality of life outcomes. This information is then reported back to the diabetes centres and relevant stakeholders to help determine what factors contribute to and promote better patient care and outcomes. The reports provide a unique opportunity for diabetes centres to benchmark their performance against their peers across Australia and the different segments of the health system. Ultimately it is hoped to be able to benchmark the performance of Australian diabetes centres with international centres.
The Australian National Diabetes Audit is fully funded by the Commonwealth Department of Health; their generous contribution and enthusiastic support for ANDA is a testament to the importance of this nationwide quality assurance activity.
Project Aims
ANDA aims to:
- Provide a diabetes dataset aligned with national reporting guidelines
- Create a centralised mechanism for providing feedback
- Benchmark clinical outcomes and improve patient care
- Identify areas of excellence and opportunities for improvement
- Assess adequacy of resources available
- Facilitate the development of procedures, clinical guidelines and policies
- Identify population groups that may require better access to care

Data Collection Modules
ANDA aims to have a broad diabetes focus and it is anticipated that the activity will expand to collect a dataset of many different aspects of diabetes care and outcomes.
Currently, ANDA has two data collection modules: Clinical indicators and Patient-reported education, self-care and quality of life indicators
Data is collected for people with diabetes attending diabetes centres in participating Australian health services.
- Clinical indicators
Clinical indicator data collection assesses a standardised set of predefined clinical parameters including demographic variables, biological variables and clinical outcomes known to impact people with diabetes.
- Patient-reported education, self-care and quality of life indicators
This data collection assesses a standardised set of predefined patient self-management parameters including lifestyle factors, education and quality of life outcomes as well as health professional utilisation.
As new modules are approved for data collection, diabetes centres will be engaged and invited to participate.
For more information about the specific type of patient information collected in ANDA, please visit our What information is collected page.
Participating Diabetes Centres
Our Supporters

Australia’s health system is world-class, supported by universal and affordable access to high quality medical, pharmaceutical and hospital services, while helping people to stay healthy through health promotion and disease prevention activities.
The Australian Department of Health has been established to provide a continued commitment of improving health outcomes for all Australians. Their purpose is to support the government and stakeholders to lead and shape Australia’s health and aged care system and sporting outcomes through evidence-based policy, well-targeted programs and best practice regulation.

The NADC is a national collective of organisations that are involved either directly or indirectly in diabetes services and care. The NADC was established to explore mechanisms and implement strategies for improving the standard of care for people with, or at risk of diabetes. NADC member organisations take a leadership role in developing the appropriate networks in their areas in order to achieve this outcome.

The ADS is the peak national medical and scientific body in Australia for diabetes. The ADS is a member-based organisation consisting of health professionals (endocrinologists, diabetes educators, primary care physicians and allied health professionals) as well as clinical and preclinical researchers. The ADS mission is to prevent diabetes and its complications; provide excellence in education for health professionals; promote collaboration, research and education; work with other organisations committed to improved diabetes treatment and care and advocate for health policies around diabetes and its prevention.
PREVIOUS ANDA FORMS, REPORTS and INFORMATION
For more information on the ANDA audits including the published reports, please click on the following links: CLICK HERE
PAST ANDA REPORTS
ANDA ARTICLES
| ARTICLES | LINK |
| Impact of COVID-19 on Diabetes Health Care and Service Provision in Australian Diabetes Centers | Click Here |
| Exploring HbA1c variation between Australian diabetes centres: The impact of centre-level and patient-level factors | Click Here |
| Making the most of audit and feedback to improve diabetes care: a qualitative study of the perspectives of Australian Diabetes Centres | Click Here |
| Age, age at diagnosis and diabetes duration are all associated with vascular complications in type 2 diabetes (Nanayakkara et al. Diabetes Complication) Journal of Diabetes and Its Complications | Click Here |
| Age-related differences in glycaemic control, cardiovascular disease risk factors and treatment in patients with type 2 diabetes: a cross-sectional study from the Australian National Diabetes Audit (Nanayakkara et al. BMJ Open) | Click Here |
| Burden of cardiovascular risk factors and disease among patients with type 1 diabetes: results of the Australian National Diabetes Audit (ANDA) (Pease et al. Cardiovascular Diabetology) | Click Here |
| Depression and diabetes distress in adults with type 2 diabetes: results from the Australian National Diabetes Audit (ANDA) 2016 (Nanayakkara et al. Scientific Reports) | Click Here |
| Younger people with Type 2 diabetes have poorer self-care practices compared with older people: results from the Australian National Diabetes Audit (Nanayakkara et al. Diabetic Medicine) | Click Here |
| The association of smoking status with glycemic control, metabolic profile and diabetic complications– Results of the Australian National Diabetes Audit (ANDA) (Szwarcbard et al J Diabetes Complications) | Click Here |
| Risk-adjustment of diabetes health outcomes improves the accuracy of performance benchmarking (Danek et al Scientific Reports) | Click Here |